× close
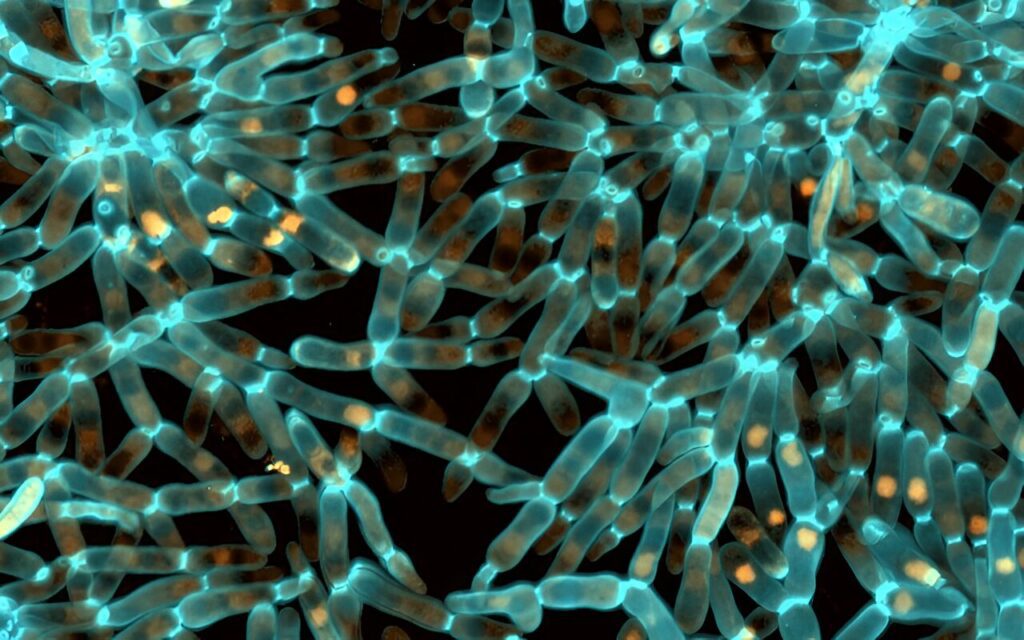
Evolved snowflake yeast.Credit: Tony Burnetti
Researchers have discovered a mechanism that guides the evolution of multicellular organisms. They identified how changes in protein folding drive multicellular evolution.
in new research Led by researchers at the University of Helsinki and the Georgia Institute of Technology, the scientists turned to a tool called experimental evolution. The ongoing Multicellular Long-Term Evolution Experiment (MuLTEE) is showing laboratory yeast evolving new multicellular functions and allowing researchers to study how they arise.
This research scientific progressfocuses on protein regulation in understanding evolution.
“By demonstrating the influence of changes in protein levels in driving evolutionary change, this study reveals why knowledge of the genetic code itself cannot fully understand how organisms acquire adaptive behaviors. Achieving such understanding requires mapping the entire flow of genetic information, “down to the operational state of proteins that ultimately control cell behavior,” says Helsinki Life Sciences Research. says Juha Saarikangas, Associate Professor at HiLIFE and the Department of Biological and Environmental Sciences at the University of Helsinki.
Snowflake yeast evolves into a robust body over 3,000 generations by changing its cell shape
One of the most important multicellular innovations is the origin of the robust body. Over 3,000 generations, these “snowflake yeasts” evolved from being weaker than gelatin to being as strong and durable as wood.
Researchers have identified a non-genetic mechanism operating at the level of protein folding that underlies this new multicellular trait. The authors found that expression of the chaperone protein Hsp90, which helps other proteins acquire functional shape, gradually declined as snowflake yeast evolved into larger and more robust bodies.
It turns out that Hsp90 functions as a crucial regulatory knob, destabilizing the central molecule that regulates cell cycle progression and causing cell elongation. This elongated shape allows cells to wrap around each other and form larger, more mechanically robust multicellular groups.
“Hsp90 has long been known to stabilize proteins and help them fold properly,” explains lead author Christopher Montrose from the Helsinki Institute of Life Sciences in Finland. . “What we discovered is that small changes in the function of Hsp90 can have profound effects not just on single cells, but on the very nature of multicellular organisms.”
Path to adaptive evolution through protein shape changes
From an evolutionary perspective, this study highlights the power of non-genetic mechanisms in rapid evolutionary change.
“We tend to focus on genetic changes, and we were very surprised to find such large changes in the behavior of chaperone proteins. This could help us solve new problems, such as building stronger bodies. “It highlights how creative and unpredictable evolution is when it comes to finding solutions,” said Will, Georgia Tech’s Ratcliffe.
For more information:
Kristopher Montrose et al, Proteostatic tuning supports the evolution of novel multicellular traits, scientific progress (2024). DOI: 10.1126/sciadv.adn2706. www.science.org/doi/10.1126/sciadv.adn2706